Medical congresses generate an enormous volume of information in a very short period of time. Sessions run in parallel, new data is presented continuously, and conversations with clinicians often add important context beyond what appears on the slides. For Medical Science Liaisons, the work does not end when the meeting concludes. In many ways, the most important work begins afterward: translating what was observed into a clear and useful post-conference report.
Yet many MSL teams find this step surprisingly difficult. A recent survey of MSL professionals highlights several common challenges in the reporting process. As shown in the figure below, the most frequently cited issue was the difficulty of reporting on all captured meeting notes (23%). Other common challenges included a lack of clear direction regarding what should be included in a conference report (17%), as well as the complexity of exporting and organizing gathered intelligence while minimizing duplication across a team (16%).
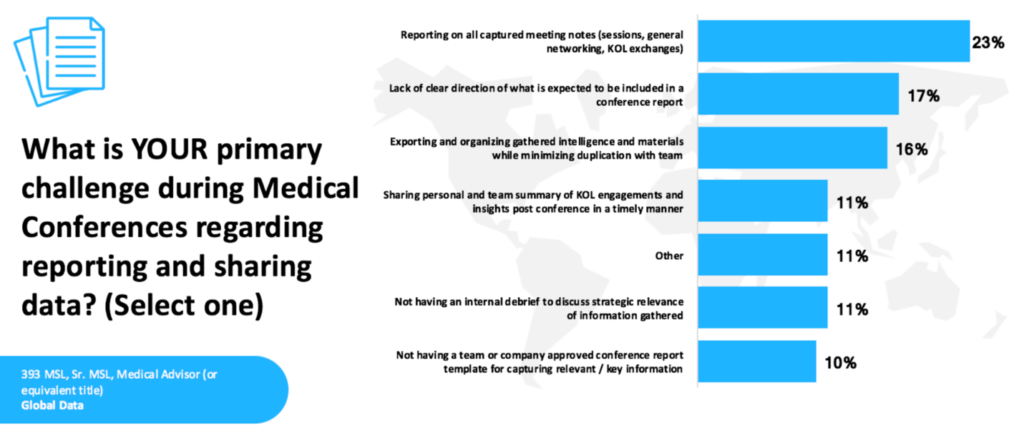
Source: The MSL Journal (https://themsljournal.com)
These results reflect something we have also seen repeatedly while supporting Medical Affairs teams covering scientific meetings. Congress coverage often produces an enormous amount of raw material—session notes, slides, conversations with clinicians, competitive observations, and internal discussions. Synthesizing that material into a concise, actionable report requires more than simply assembling notes after the meeting.
The most effective MSL teams approach the post-conference report differently. Instead of attempting to document everything that happened during the congress, they focus on distilling the information that will actually influence decisions inside their organization.
Below are several practices we consistently see among teams whose post-conference reports become valuable internal resources rather than administrative exercises.
Start With the Information That Matters Most
A common reporting mistake is attempting to document every presentation attended. While comprehensive documentation may feel thorough, it often produces reports that are lengthy but difficult to interpret.
High-value reports prioritize the information most likely to influence clinical strategy, research planning, or scientific engagement.
This may include major clinical findings, notable reactions from respected clinicians, or emerging themes that signal a shift in the treatment landscape. The goal is clarity, not volume.
When internal teams read the report, they should be able to quickly identify the developments that matter most.
Explain Why the Insight Matters
Raw observations rarely provide enough context for internal stakeholders. A useful report explains not only what was observed, but why it matters.
For example, a new study result becomes significantly more valuable when the report clarifies how it may influence clinical practice, future research directions, or competitive positioning.
Strong post-conference reports therefore connect observations to implications. They help internal teams understand what the information means and what actions may follow.
Structure the Report for Busy Stakeholders
Many stakeholders reviewing congress reports have limited time. A well-structured document allows readers to understand the most important findings quickly without working through pages of detail.
The most effective reports begin with a concise executive summary. This section highlights the key insights, major scientific developments, and the overall significance of the meeting. For many readers, this summary will provide the primary value of the report.
The body of the report can then provide additional context. This may include observations from sessions, summaries of KOL discussions, competitive intelligence, and interpretation of how the findings may influence the therapeutic landscape.
When the structure is clear, internal teams can quickly navigate the sections most relevant to their work.
Capture Context From Conversations
Some of the most valuable insights from a congress do not come directly from the slides presented on stage. They emerge from conversations with clinicians, informal discussions after sessions, and reactions from experts evaluating new data in real time.
Documenting these perspectives can add depth to a report that pure session summaries cannot provide.
A brief observation from a respected clinician can help explain how the field is interpreting new findings. These perspectives often reveal how data may influence treatment decisions long before formal guidelines are updated.
Translate Observations Into Strategic Insight
The strongest post-conference reports go beyond summarizing events. They interpret what the information may mean for the organization.
This often involves identifying themes that appeared repeatedly throughout the meeting, recognizing where the scientific conversation is moving, and highlighting developments that could influence clinical development or medical strategy.
In some cases, the most valuable part of the report is the interpretation itself: explaining why a particular finding, discussion, or debate may shape the therapeutic landscape over time.
Another perspective that experienced teams often consider is how the new information compares with what was presented at previous meetings. Many congress updates represent the next step in an ongoing scientific story rather than a completely new development. Looking back at prior congress coverage can help teams quickly determine whether a presentation reflects a meaningful shift in evidence or simply an incremental update to findings already discussed the year before. Having this historical context often makes it much easier to interpret the significance of new data as it emerges.
Capture Information While the Meeting Is Still Fresh
One reason post-conference reporting becomes difficult is that too much work is postponed until after the meeting ends.
Experienced teams document key takeaways during the congress itself. Short notes written immediately after a session or discussion often preserve context that would otherwise be difficult to reconstruct later.
Many teams also conduct short daily debriefs during the meeting. These conversations allow team members to compare observations, identify emerging themes, and ensure that important insights are not overlooked.
By the time the conference concludes, much of the thinking behind the final report has already taken shape.
Reducing Friction During the Reporting Process
Across the congress coverage projects we support, one operational challenge appears frequently: assembling slides, notes, photos, and commentary into a single coherent report after the meeting.
Many organizations still manage this process with spreadsheets, shared drives, and personal note systems. These approaches can work, but they often introduce unnecessary friction when teams try to consolidate their materials afterward.
This experience is one of the reasons we developed the inVision platform at inThought. The system was originally built to support our own analysts covering complex medical meetings.
Slides can be attached directly to the sessions where they were presented. Notes and analysis remain linked to the relevant context. Captured images are automatically indexed so the embedded text becomes searchable later.
When information is captured in a structured way during the meeting, the post-conference report becomes much easier to assemble. Instead of reconstructing what happened, teams can focus their attention on interpreting what the science actually means.
Final Perspective
The post-conference report is more than a documentation exercise. It is one of the primary ways organizations translate congress observations into institutional knowledge.
When the report focuses on high-value insights, provides clear interpretation, and is structured for busy stakeholders, it becomes far more than a summary of events. It becomes a resource that helps internal teams understand how the scientific conversation is evolving.
If improving the way your organization captures and interprets congress insights is on the agenda this year, feel free to reach out, we are always happy to share what we have learned from supporting teams in the field.